Life – Terror. Ecstasy. Fight. Denial. Flight. Failure. PAIN. Forgiveness. Reconciliation. Hope. Love. Peace – Death.
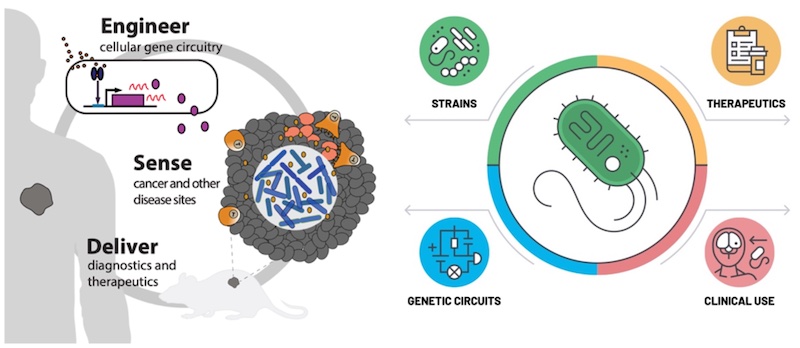
Can bacteria be engineered in ways that they help our bodies better fight cancer? That’s the goal Tal Danino, Ph.D., and The Cancer Research Institute (CRI) ‘Old STAR’, research program, named after the “Father of Modern Tumour Immunology.”
Trillions of bacteria naturally reside in and on our bodies.
Bacteria perform important functions, and without them we wouldn’t survive. On the skin and in the gut, they help regulate the immune system and keep its activity balanced.
Many people might not be aware that bacteria have anything to do with cancer, let alone that bacteria can be modified to have anti-cancer properties. There are three fields that fundamentally fit into this new approach of engineering bacteria as a cancer therapy.
The first field is microbiology, which recently has changed not only our science but our culture. We now understand that bacteria can be good as well as bad.
There are thousands of different species of bacteria in the human body and collectively, along with viruses, fungi, and other microscopic organisms, they form the microbiome.
Importantly, these bacteria interact with our cells and are needed to help the body carry out many important functions. The impact of microbiology research has equipped us with the necessary tools to discover, characterize, and understand the roles that bacteria play in human health. This could potentially build on the idea that we could change our microbiome through diet or with probiotics for not only improving health but also curing diseases such as cancer.
Thus, the concept of using bacteria as a therapy has challenged the scientific dogma of how they were viewed as “all bad” over the past decades – even after 100 years ago during the time of William Coley, when he was the first to use bacteria to treat inoperable sarcomas, an aggressive form of cancer that grow in connective tissue like bones, nerves, muscles, tendons, cartilage, and blood vessels.
Not only are bacteria found throughout our bodies, they’re also found inside people’s tumours, and scientists are now sequencing tumours to characterize these bacteria to determine what roles they play during cancer development.
The second important field is Cancer Immunotherapy (see previous posts). Using bacteria in the first cancer immunotherapies. With the 2018 Nobel Prize being awarded to Dr. James Allison, the director of the CRI Scientific Advisory Council, cancer immunotherapy has now been cemented as the fourth pillar of cancer treatment.
The third field, which has been most important is synthetic biology. In particular, using synthetic biology to engineer living cells and bacteria.
More researchers have started to utilize technology to study biological systems, like cancer or bacteria, synthetic biology, trying to learn how to re-program bacteria and build what we call genetic circuits that could control their behaviour, both of individual bacteria and of bacterial communities.
As things have progressed, to focus more on the potential applications for patients, to think about how we could merge our knowledge of bacteria and our ability to program them in ways that enable us to diagnose or even treat cancer.
William Coley as well as the work of Lloyd Old, who showed that bacteria could be used to treat bladder cancer. Their research actually contributed to the first FDA-approved immunotherapy in 1990, which uses a weakened strain of bacteria.
There’s one component to how our immune system interacts with bacteria that’s really unique in the context of cancer, which is that the cores of many tumours are immuno-privileged. What that means is that the immune system doesn’t really have much access to the inner regions of the tumour. Essentially, bacteria can use the core as a safe haven to hide. If they were anywhere else, especially in the blood, they would be eliminated.
Another place that harbours bacteria in a unique fashion is the gut.
Our intestinal tract is home to a lot of diverse bacteria, many beneficial and necessary. In addition to helping us digest food, these bacteria interact with and help educate the immune system in complex ways.
They can also impact cancer and how it responds to treatment, both with chemotherapy and immunotherapy. The fact that bacteria in your gut can affect how melanoma on the skin responds to treatment is such a fascinating concept,
There’s a long history behind our current knowledge of how bacteria naturally interact with our immune system and how that impacts how we treat cancer. To take it a step further and customize bacteria to make them better at stimulating the immune system to go after cancer is the next step. Using gene circuits to reprogram bacteria.
The history of synthetic biology is interesting, we’ve been using recombinant DNA and genetic engineering technologies in bacteria since the 1970s to produce different kinds of human proteins, such as insulin for people with diabetes.
But for a while, genetic engineering didn’t incorporate a lot of what we think about as the engineering approach, which revolves around the concept of design-build-test. A cyclical process to design systems, build them and then test them. To build off what worked and get rid of what didn’t. Much of this process revolves around mathematical models, sense and respond concept.
This requires using synthetic biology to build a genetic circuit, such as within the genome of a bacterium. Then, one component of the circuit is designed to sense whether or not it’s in a tumour environment. If—and only if—it senses that it is within a tumour, it triggers another component of the circuit to start to make a drug or do other things that would be detrimental to the tumour and beneficial for the patient.
Beyond sense and respond, the new paradigm is trying to engineer living medicines, similar to CAR T cells and other cell therapy-based approaches. It’s interesting to compare them to bacteria, because whereas CAR T cells have not had much success when it comes to infiltrating and attacking solid tumours, bacteria readily penetrate these cancers.
In this regard, they have a different role as a therapeutic modality. The future is not just going to be focused on bacteria, but on how you can get all these different modalities to start interacting with and synergizing with one another.
Even with how effective immunotherapy is in some types of advanced cancer, it still doesn’t work for the majority of patients, and it’s going to take different combinations of approaches to cure everyone.
STAR is using engineered bacteria as ‘trojan horses’ to infiltrate and undermine tumours from within. Once there, they can deliver drugs, such as chemotherapies that stop the tumour from growing, or immunotherapies that boost the immune system.
The exciting part of this approach is that bacteria colonize solid tumours regardless of their genetics. This is the key that differentiates this approach from other technologies. Being able to penetrate the low-oxygen, immune-privileged cores of solid tumours is huge, because solid tumours account for more than 90% of cancer-related cases and deaths.
This unique feature allows a get in “for free” approach, in a way, without having to engineer that capability into the bacteria. They can colonize the tumour core, the deepest and most difficult region to target with other types of drugs.
Bacteria can act on the deepest parts of the tumour, and other drugs act on the proliferating outer parts of the tumour. For this reason alone, bacteria are a really interesting modality to combine with other therapies.
PD-1/PD-L1 checkpoint immunotherapies, which are probably the most broadly successful immunotherapies today. work by preventing tumours from shutting down ongoing immune responses, and they’re most effective against tumours that have already been infiltrated by T cells that recognize the cancer cells. But in most cases, tumours either exclude these T cells or lack them altogether, and this renders PD-1 immunotherapy ineffective.
For these hard-to-treat tumours, there is potential to engineer bacteria to produce molecules that attract the immune system, and thereby trigger immune responses from scratch and enhance the power of immunotherapy? A cytokine called GM-CSF can recruit T cells and other immune cells. And in “colder” tumours that are lacking in immune cells, it had a stronger effect in combination with checkpoint immunotherapy molecules produced by the bacteria. So those together was more effective than one or the other.
The next steps
Identify some promising bacterial therapy candidates through translational studies so that we could then advance to clinical trials., some of which have already started.
The hope is to develop new candidates with even better effectiveness.
Thanks For Reading
Hope
Cancer Immunotherapy: 2022 Research https://www.cancerresearch.org/events/webinars/cancer-immunotherapy-2022-research-john-wherry?utm_source=luminate&utm_medium=email&utm_campaign=cancerimmunews&utm_content=feb2022